Saturday, April 16, 2011
[Breaking] Indications of Fukushima Iodine 134 Contamination in Saint Louis
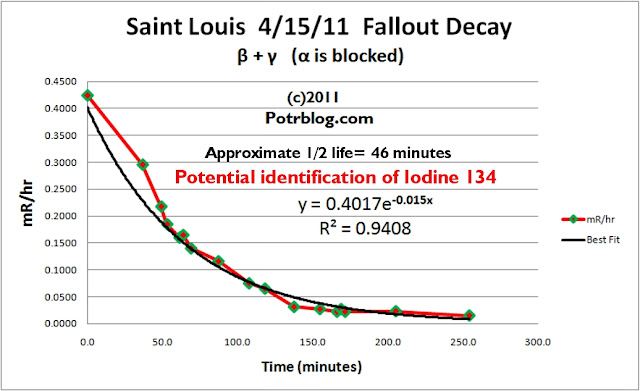
The above chart is of data recorded from the Saint Louis 4/15/11 radioactive thunderstorm fallout sample which was wiped from the nose/grill of my SUV.
The data was taken from a sample sealed in a Ziploc Vacuum bag.
The sample was sealed in the vacuum bag to prevent potential radioactive dissipation from off-gassing.
The data was right censored (after 255 minutes) to improve the signal to noise ratio as the decay approached background levels.
The half life was calculated from the exponential trend line fit of the empirical data.
The resultant half life was calculated to be approximately 46 minutes.
Given the probabilistic nature of decay and the trend line fit error, the calculated half life can not be taken as an exact value.
Radioactive decay chains were examined for beta decay isotopes with half lives falling within +/- 20 minutes of the calculated 46 minute half life.
A cursory examination was made of isotope decay chains whose superposition of half lives might approximate a 46 minutes half life.
An examination was made of reported Fukushima fission release products and possible daughters with the requisite half life.
Based on the empirical data analysis and reports of massive releases of I134 (and parent), a strong potential exists that the radioactive fallout contamination sample collected here in Saint Louis Missouri is Iodine 134.
Given the fleeting detectable nature of I134 fallout, and the potential risk to the human thyroid, further vigilance and analysis should be performed by a qualified laboratory.
STAY OUT OF THE RAIN!
Labels:
Fukushima,
radioactive
Subscribe to:
Post Comments (Atom)
Hello there,
ReplyDeleteI've been following your blog for weeks and have posted here my questions from time to time.
I'm still left with the question, why my PM1703M is not picking up ANY activity for the past 3 weeks. I have done the same thing with a paper towel picking up rain from my car and tested several rain puddles in the Oregon area and I get absolutely no change. I know the device is working because I do in fact get activity from a banana, brick buildings and granite. I'm not questioning your blog or the videos but I'm just trying to figure this all out.
Christine,
ReplyDeleteNothing wrong with questions. Your detector only detects Gamma radiation. The fallout I have detected here in Saint Louis has primarily been a Beta emitter. That means your detector would be unable to detect it.
There are some additional issues; your detector was designed for first responders and customs officials to detect weapons material smuggling activity. In that regard, your meter has a very compact sensor. It was not designed to test for food/ surface contamination.
One final aspect is that it is more difficult to pick up detections through water / liquids. Your best bet to detect something is to leave a piece of sheet metal (or your car) in your yard; after a rain shower, wipe the sheet metal dry with a 2 paper towels (don't soak them); then very slowly move the detector over the paper towel and look for hot spots. You can also try the same test by wiping down the hood of your car. Again, if there is only alpha and beta radiation your detector won't detect it.
Interestingly enough, there seem to be some rumblings on the grapevines that EPA's RADNET is having failures or blackouts with Beta readings.
Ms. X ~ Thank you for this explanation. I'm not going to feel better until I have a detection device that I can use on food and surface. I need to protect my children! It's very hard to do that with out "knowing" so to speak. Is there a device that detects all three (gamma, alpha, beta) or do I need to get two separate devices? I'm getting to the point where I just want to urge my husband to quit his job, get passports for the kids and leave the Northern Hemisphere. I know this seems extreme but gosh darn I'm sick of not knowing what is harming them.
ReplyDeleteChristine,
ReplyDeleteThe medcom inspector we have does detect all three types of radiation, but the back order on them is up into months now.
In regards to leaving the hemisphere, the president and his family did that when the event first impacted the USA. And given unlimited funds this would be a good time for an extended vacation south of the equator (or at least lay the ground work to rapidly do so).
But as far as quitting the jobs and cutting to run, there is a much larger threat picture (made worse by Fukushima) to mitigate, involving economics, limited global resources, unlimited printing of money, warfare and authoritarianism. My family's solution to deal with the uncertainties is to get on our knees every evening and pray.